Redefining Cancer Care
We are channeling the power of mitochondrial dynamics to address unmet needs in oncology
At Bantam Pharmaceutical, we are developing novel, first-in-class small molecule oral therapeutics for difficult-to-treat hematological and solid tumors.
Our mission is to destroy cancer cells from the inside, using our unique expertise to alter their inner mitochondrial dynamics.
Our Science
In the fight against cancer, Bantam Pharmaceutical is taking a new approach.
We have developed a novel mechanism to target and kill cancer cells.
One of the hallmarks of cancer cells are their ability to resist apoptosis (cell death). Our technology is able to restore apoptosis and generate anti-tumor effects.
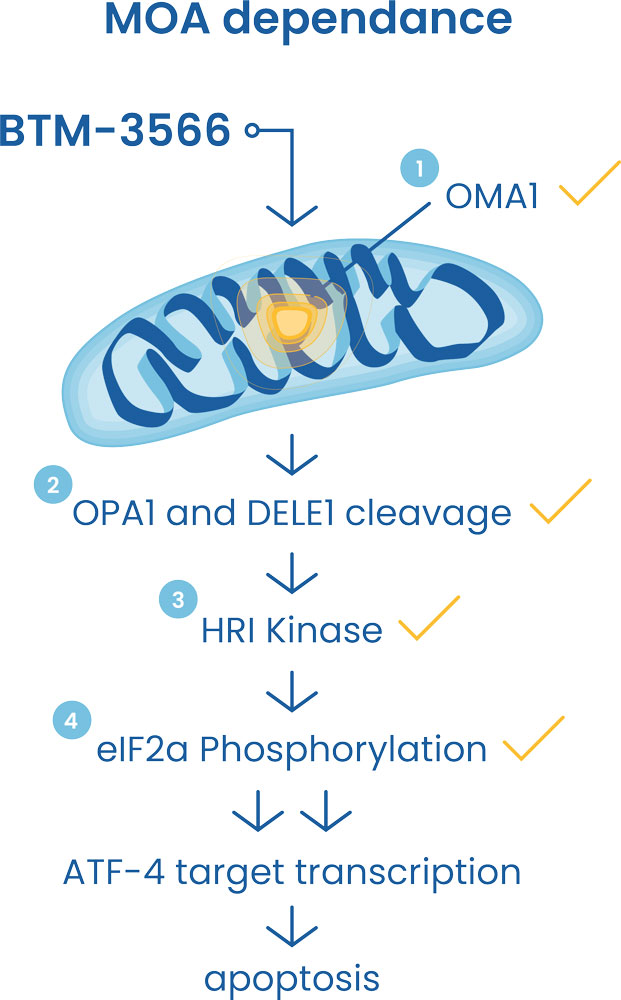
Our lead candidate is BTM-3566, an orally-available novel small molecule compound with broad anti-cancer activity in hematologic and solid tumors, initially focused on Diffuse Large B-cell Lymphoma (DLBCL).
In preclinical work to date, BTM-3566 has shown exceptional in vitro and in vivo activity against DLBCL, inducing durable and complete regression in primary tumor models.
We believe BTM-3566 could revolutionize how we treat cancer, and ultimately save lives.
Our People
OUR TEAM
Our team has extensive experience in oncology drug discovery and development from bench to market.
OUR BOARD
Accomplished scientific advisory board comprised of experienced academic and industry leaders.
OUR MODEL
Capital efficient operating model has delivered value and is poised for aggressive product development.
We are an early mover in the field of mitochondrial homeostasis, using our pioneering science and unrivaled expertise in mitochondrial cellular biology to revolutionize oncology.
Capital efficient operating model has delivered value and is poised for aggressive product development.
